Getting Started
As a Principal Investigator, you are responsible for performing research with experimental animals safely and completing all required safety forms.
Register your lab in Lumen
Lumen is the University’s online safety data management tool used by Principal Investigators to submit applications and registrations for review.
To register your lab in Lumen, follow the instructions here.
Once registered in Lumen, complete the Laboratory Setup process, which collects information on research focus, hazards and job activities, and lab members.
Complete your Biological Registration in eIBC
The Institutional Biosafety Committee uses eIBC to review and approve research involving recombinant and synthetic nucleic acids. Hazardous agents that must be described in your eIBC submission include viral vectors, recombinant and wild-type viruses, other recombinant or wild-type pathogens, and biological toxins. See the eIBC page for more information.
Research Safety assists IACUC by performing a risk assessment of the research in the Animal Study Protocol (ASP).
Research Safety provides safety guidelines for handling chemical, biological and radiological hazards used in the ASP (Occupational Health and Safety section). The review process ensures that the ASP includes safety guidelines for all hazardous or potentially hazardous materials and procedures at appropriate biosafety levels.
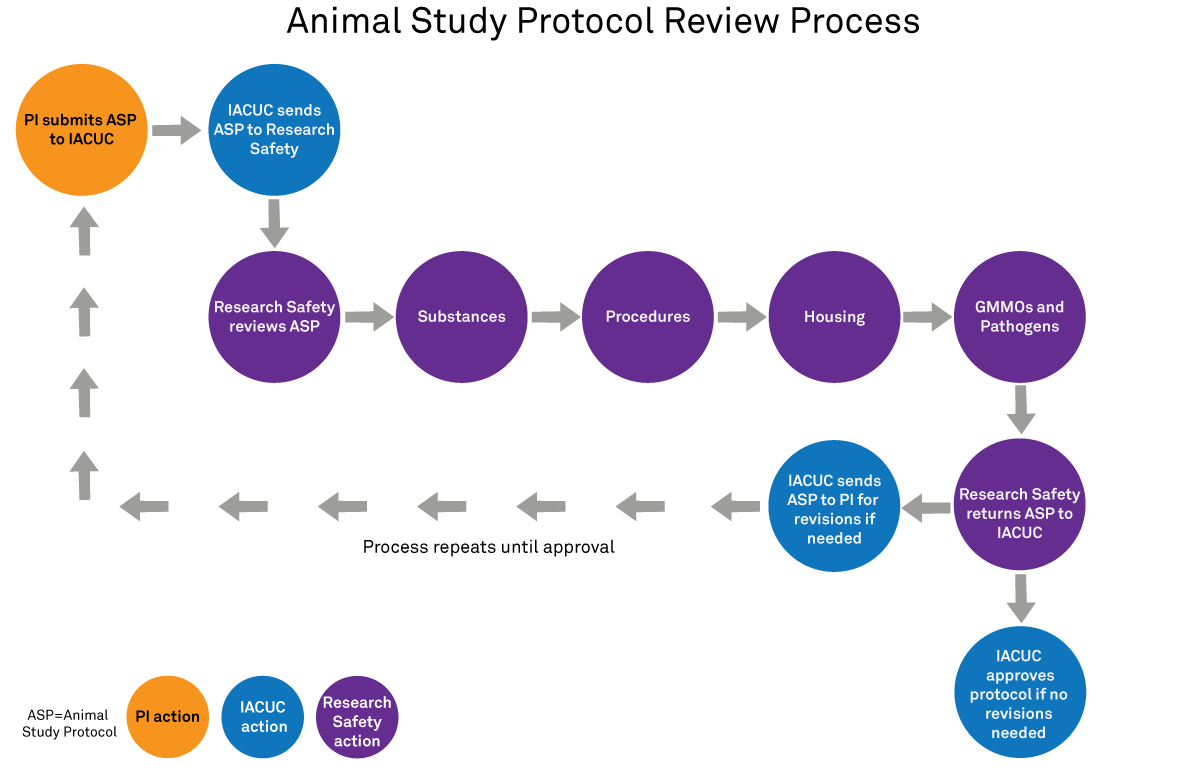
If you need assistance completing the Occupational Health and Safety section of the ASP, contact us at animalresearchsafety@northwestern.edu.
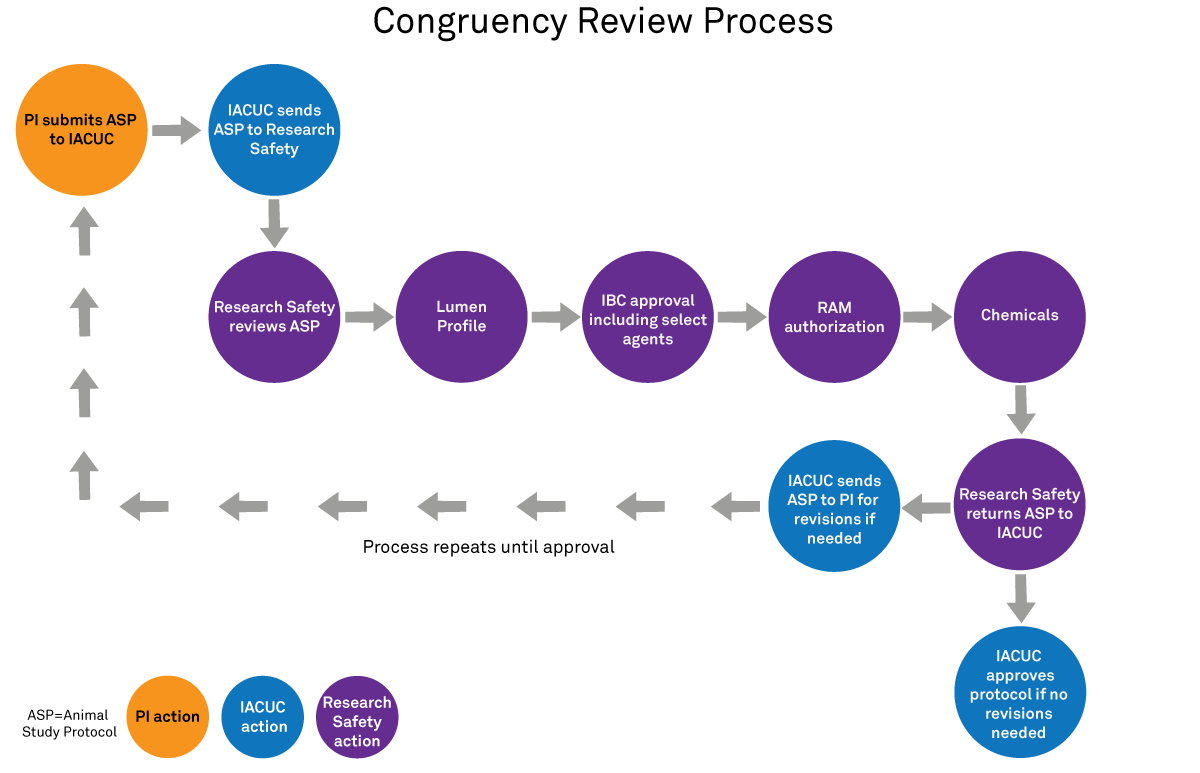
During the IACUC review process, Research Safety conducts a congruency review of the Animal Study Protocol (ASP) submission. Research Safety ensures that:
- The Lumen profile is complete (all required safety forms and trainings are complete)
- The IBC has approved Biological Registration
- The use of radioactive materials (RAM) is authorized
- The use of hazardous (or potentially hazardous) chemicals is approved
Emergency approval of the ASP may be granted (for example, to meet a grant proposal deadline); however, animal work may not be performed prior to IBC approval or RAM authorization.
The Principal Investigator and all lab members listed on an Animal Study Protocol are required to complete safety training provided by Research Safety, as well as any training required by IACUC and CCM.
Research Safety training is assigned through Lumen and completed in myHR Learn. To view your training requirements in Lumen, follow these steps:
- Log in to Lumen
- Select Training (in the left menu)
- Select My Training (in the left menu)
- Select the Training Requirements tab
Visit the IACUC training page to determine what training is required by IACUC. IACUC training is delivered through the AALAS Learning Library.
Visit the CCM Onboarding for New Research Personnel page to determine what training is required by CCM and how to register.
For additional questions regarding safety training, email safety-training@northwestern.edu.